当Ni以粒子/纤维形式弥散分布于Ag基体时,Ag-Ni合金具有高导电、高导热、高比热、耐电磨损等性能,并能在大气环境下保持低而稳定的接触电阻,是优良的电接触材料,目前广泛应用于中低负荷接触器、断路器、继电器等[1,2]。Ag-Ni是典型的偏晶合金,具有组元液态不混溶区间。由Ag-Ni相图[3]可知,过偏晶成分的银基Ag-Ni合金熔体在冷却过程中先后发生液-液相变、弥散相(富Ni相)液滴凝固、弥散相粒子沉淀析出和成分接近于纯Ag的基体熔体凝固等过程,易于形成偏析严重的凝固组织[4]。迄今为止,对Ag-Ni合金凝固过程的研究极少[5,6,7],工业上通常用粉末冶金技术制备Ag-Ni合金(Ni含量≥5%,质量分数)电接触材料。然而,用粉末冶金法制备的Ag-Ni合金Ni相粒子尺寸较大、易发生团聚,强化作用较弱,需要较高的Ni含量才能达到满意的强化效果。
近年来,人们对偏晶合金凝固过程开展了大量的实验[8,9,10,11,12,13,14,15,16,17,18,19]和模拟研究[20,21,22,23,24,25,26,27,28],结果表明,弥散相液滴/粒子的形核特性和空间迁移特性是决定偏晶合金能否形成弥散型凝固组织的关键因素。快速、亚快速凝固能大幅度提高弥散相液滴/粒子的形核速率、减小弥散相液滴/粒子的尺寸,缩短合金熔体通过液-液相变温度区间的时间,因此,能有效抑制相偏析,促进弥散型凝固组织形成,在偏晶合金制备上具有很好的应用前景。据此,本工作对Ag-Ni合金开展了快速/亚快速凝固实验,建立了描述Ag-Ni合金凝固过程的理论模型,将实验与模拟相结合,研究了Ag-Ni合金凝固过程及合金成分、试样直径(冷却速率)对凝固组织的影响。
1 实验方法
以纯度为99.99% (质量分数,下同)的Ag和Ni为原料,按不同成分Ag-Ni合金进行配料,得到Ag-1.25%Ni、Ag-2.24%Ni、Ag-3.0%Ni和Ag-4.0%Ni合金;然后将配制好的金属原料装入石墨坩埚内,在SPG50K-25B型高频感应炉中加热熔化并升温至2023 K;保温10 min,期间充分搅拌合金熔体以保证形成均一熔体;最后将合金熔体浇入预热至423 K的石墨模中冷却凝固,得到直径分别为4、5、6和8 mm,高为60 mm的圆柱形试样。实验过程中采用XSR90-04V0型彩色无纸记录仪搭配直径为0.1 mm的W-Re热电偶测定试样中心的冷却曲线,记录时间间隔为0.1 s。实验后沿轴向切割样品,对切面进行研磨、抛光,制备金相试样,利用Quanta 450型扫描电子显微镜(SEM)观察显微组织,采用SISC IAS V8.0定量金相图像分析软件测定富Ni相粒子的尺寸分布和平均半径。利用401MVD型显微Vickers硬度计测量试样剖面硬度,根据GB/T 5586-2016确定载荷为2.94 N,保持时间为30 s。
2 实验结果
图1为不同直径Ag-4.0%Ni合金试样心部的冷却曲线。由图可见,试样越大,其冷却速率越小。
图2给出了直径为8 mm的Ag-4.0%Ni合金样品中富Ni相粒子分布图及其体积分数沿试样轴向和径向的分布。可见,富Ni相粒子体积分数在试样两端和表面随位置变化较大,而在试样内部区域基本呈均匀分布。
图1
图1
不同直径(d) Ag-4.0%Ni合金试样心部的冷却曲线
Fig.1
Experimental results (symbols) and calculated results (solid lines) of the cooling curves for the center region of Ag-4.0%Ni alloys with different diameters (d) (T—temperature, t—time, Tm and Te refer to the monotectic reaction temperature and the eutectic reaction temperature of Ag-Ni alloy, respectively)
图2
图2
直径为8 mm的Ag-4.0%Ni合金中富Ni相粒子分布示意图及实测富Ni相粒子体积分数(
Fig.2
Schematic distribution of the Ni-rich particles in Ag-4.0%Ni alloy with a diameter of 8 mm (a), measured volume fraction (
图3
图3
直径为5 mm的Ag-xNi合金显微组织的SEM像
Fig.3
SEM images of the Ag-xNi alloys for the samples of 5 mm in diameter with x=1.25% (a), x=2.24% (b), x=3.0% (c) and x=4.0% (d)
Fig.4
Fig.4
2D size distributions of the Ni-rich particles in Ag-xNi alloys for the samples of 5 mm in diameter with x=1.25% (a), x=2.24% (b), x=3.0% (c) and x=4.0% (d)
图4直径为5 mm的Ag-xNi合金中富Ni相粒子的二维尺寸分布
图5
图5
Ag-Ni合金中富Ni相粒子二维平均半径(<R>2D)随合金成分的变化
Fig.5
Average 2D radius (<R>2D) of the Ni-rich particles in Ag-Ni alloys for the samples of 5 mm in diameter vs alloy composition
图6
图6
不同直径Ag-4.0%Ni合金试样凝固组织的SEM像
Fig.6
SEM images showing solidification micro-structures of Ag-4.0%Ni alloys with diameters of 4 mm (a), 6 mm (b) and 8 mm (c)
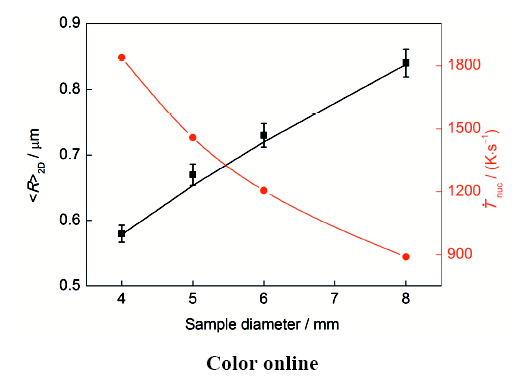
图7
图7
Ag-4.0%Ni合金中富Ni相粒子<R>2D和富Ni相液滴形核阶段熔体冷却速率(
Fig.7
Experimental results (black symbols) and calculated results (black line) of <R>2D of the Ni-rich particles in Ag-4.0%Ni alloys and the cooling rate of the melt during the nucleation (
图8为实验测得的Ag-Ni合金显微硬度随合金成分和试样直径的变化。可见,随着合金Ni含量的增加以及试样直径减小,Ag-Ni合金显微硬度逐渐增大,其中,直径为4 mm的Ag-4.0%Ni合金显微硬度可达48.13 HV,接近粉末冶金生产的Ag-10.0%Ni片状电触头的硬度(50 HV)。
图8
图8
Ag-Ni合金显微硬度随合金成分和试样直径的变化
Fig.8
Microhardnesses of Ag-Ni alloys as a function of alloy composition for the samples of 5 mm in diameter (a) and as a function of sample diameter for the Ag-4.0%Ni alloys (b)
3 分析讨论
3.1 凝固过程的模型构建
式中,uM为富Ni相液滴的Marangoni运动速率;vi为富Ni相液滴/粒子的长大速率;
式中,N0为单位体积熔体内原子数;nci为具有临界晶核半径
合金试样冷却过程中温度场满足如下方程[25]:
式中,kmix、ρmix和
合金凝固过程中,溶质主要通过扩散以及富Ni相液滴空间迁移进行传输。根据溶质守恒关系,熔体内的浓度场满足如下方程[25]:
式中,
模拟计算中,Ag基体熔体的动力学黏度(η)用下式计算[31]:
式中,NA为Avogadro常数。
D通过Sutherland-Einstein关系式计算[32]:
式中,rNi为Ni的原子半径。
偏晶合金两液相间界面能(σL-L)用下式计算[29]:
式中,σ0为常数,TC=2747 K为Ag-Ni合金体系的临界温度。
| Parameter | Value | Unit |
|---|---|---|
| Thermal conductivity of liquid Ag | 122.29093+0.04259T | W·K-1·m-1 |
| Thermal conductivity of liquid Ni | 57 | W·K-1·m-1 |
| Thermal conductivity of solid Ag | 429 | W·K-1·m-1 |
| Thermal conductivity of solid Ni | 90.7 | W·K-1·m-1 |
| Density of liquid Ag | 9330-0.91(T-1233.7) | kg·m-3 |
| Density of liquid Ni | 7905-1.19(T-1727) | kg·m-3 |
| Density of solid Ag | 10500 | kg·m-3 |
| Density of solid Ni | 8900 | kg·m-3 |
| Specific heat of liquid Ag | 283 | J·kg-1·K-1 |
| Specific heat of liquid Ni | 620 | J·kg-1·K-1 |
| Specific heat of solid Ag | 235 | J·kg-1·K-1 |
| Specific heat of solid Ni | 444 | J·kg-1·K-1 |
| Latent heat of solidification of pure Ag | 102809 | J·kg-1 |
| Latent heat of solidification of pure Ni | 292334 | J·kg-1 |
3.2 模拟分析
通过对合金凝固组织进行拟合计算来确定富Ni相粒子与基体熔体之间的界面能(σP-L)及σL-L。即首先选取不同的σP-L模拟计算凝固过程中只发生富Ni相粒子沉淀析出的直径为5 mm的亚偏晶Ag-2.24%Ni合金试样凝固组织形成过程,通过对比模拟结果与实验结果,确定σP-L=0.083 J/m2;然后应用该σP-L,并选取不同的σ0模拟计算凝固过程中同时存在液-液相变和富Ni相粒子沉淀析出的直径为5 mm的过偏晶Ag-4.0%Ni合金试样凝固组织形成过程,通过对比模拟结果与实验结果,确定σ0=0.25;进而利用上述界面能计算了不同成分、不同直径Ag-Ni合金试样的凝固过程,模拟给出的富Ni相粒子的二维平均尺寸如图5和7所示。模拟结果和实验统计结果相符,进一步验证了界面能取值的正确性,也说明所建模型能描述Ag-Ni合金凝固组织演变过程。
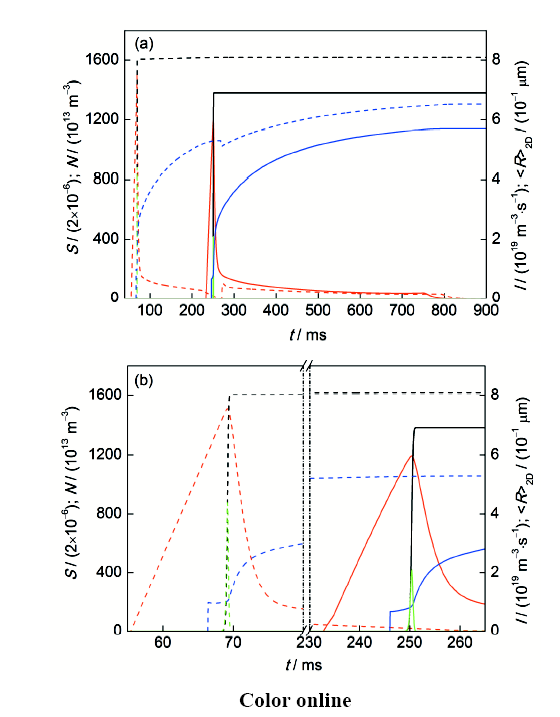
图9给出了直径为5 mm的亚偏晶Ag-2.24%Ni和过偏晶Ag-4.0%Ni合金试样凝固过程中试样心部基体熔体过饱和度,富Ni相液滴/粒子形核率、数量密度和二维平均半径随凝固时间的变化曲线。可见,当Ag-2.24%Ni合金熔体冷却进入液-固相区后,随着温度的降低,熔体过饱和度逐渐增加。当过饱和度达到某一临界值时,富Ni相粒子开始形核。在形核初期,富Ni相粒子的形核率和数量密度较低,富Ni相粒子形核和长大导致的过饱和度下降不足以抵消熔体冷却导致的过饱和度上升,因而熔体过饱和度增加,富Ni相粒子形核率和数量密度迅速提高。不久后,富Ni相粒子形核和长大主导过饱和度变化,熔体过饱和度逐渐下降,形核停止,富Ni相粒子在冷却过程中继续长大。Ag-4.0%Ni合金熔体冷却凝固过程中首先发生液-液相变,富Ni相液滴的形核、长大过程与Ag-2.24%Ni合金凝固时富Ni相粒子的形核、长大过程相似。当熔体温度降至偏晶反应温度时,富Ni相液滴发生偏晶反应,转化为富Ni相粒子,其平均尺寸略微减小,此后,在富Ni相粒子长大作用下基体熔体过饱和度始终较低,熔体内不再发生富Ni相粒子的形核。
图9
图9
Ag-2.24%Ni和Ag-4.0%Ni合金试样心部基体过饱和度,富Ni相液滴/粒子形核率、数量密度和二维平均半径随凝固时间的变化曲线
Fig.9
Supersaturation (S, red line) of the matrix melt, nucleation rate (I, green line), number density (N, black line) and <R>2D (blue line) of the Ni-rich droplets/particles in the center region of the sample as a function of time during cooling the Ag-2.24%Ni alloy (solid line) and Ag-4.0%Ni alloy (dashed line) for the samples of 5 mm in diameter (a), and the enlargement of the microstructure evolution during the period from 55 ms to 265 ms (b)
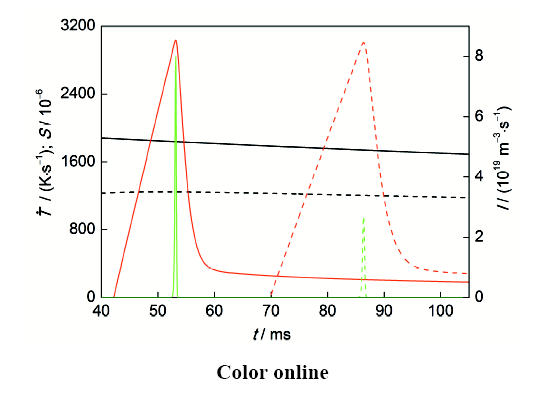
图10
图10
不同直径Ag-4.0%Ni合金试样冷却时试样心部冷却速率、基体熔体过饱和度和富Ni相液滴形核率随凝固时间的变化曲线
Fig.10
Cooling rate (
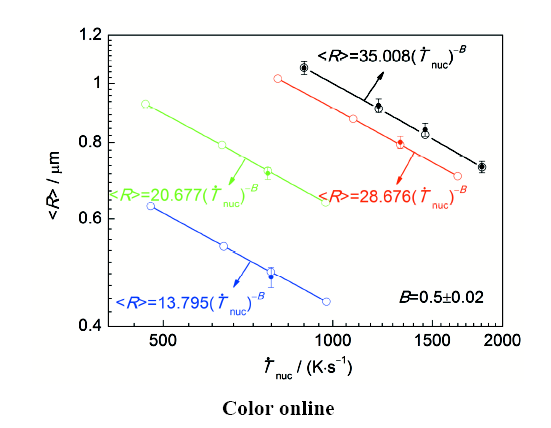
图11
图11
不同成分Ag-Ni合金凝固组织中富Ni相粒子平均半径(<R>)随富Ni相液滴/粒子
Fig.11
Experimental results (solid circles) and calculated results (open circles) of the average radius (<R>) of the Ni-rich particles in Ag-xNi alloys with x=1.25% (blue color), x=2.24% (green color), x=3.0% (red color), x=4.0% (black color) vs
4 结论
(1) 利用快速/亚快速凝固方法可制备富Ni相粒子弥散分布于Ag基体的Ag-Ni合金。
(2) Ag-Ni合金显微硬度随着合金Ni含量增加和试样凝固过程冷却速率升高而增大。当Ag-4.0%Ni合金液-液相变开始阶段熔体冷却速率达1800 K/s时,其显微硬度为48.13 HV,接近于粉末冶金生产的Ag-10.0%Ni的硬度。快速/亚快速凝固在高性能Ag-Ni触头材料制备上具有很好的应用前景。
(3) Ag-Ni合金的Ni含量越高、凝固过程中冷却速率越低,则凝固组织中富Ni相粒子平均尺寸越大。富Ni相粒子的平均半径(<R>)与富Ni相液滴/粒子形核阶段熔体冷却速率(
(4) 在快速/亚快速凝固条件下,Ag-Ni合金凝固过程中富Ni相液滴/粒子的Ostwald粗化作用很弱,富Ni相液滴/粒子的尺寸主要受形核和长大控制。